Influence of Process Parameters on Content Uniformity of a Low Dose Active Pharmaceutical Ingredient in a Tablet Formulation According to GMP – topic of research paper in Chemical sciences. Download scholarly article
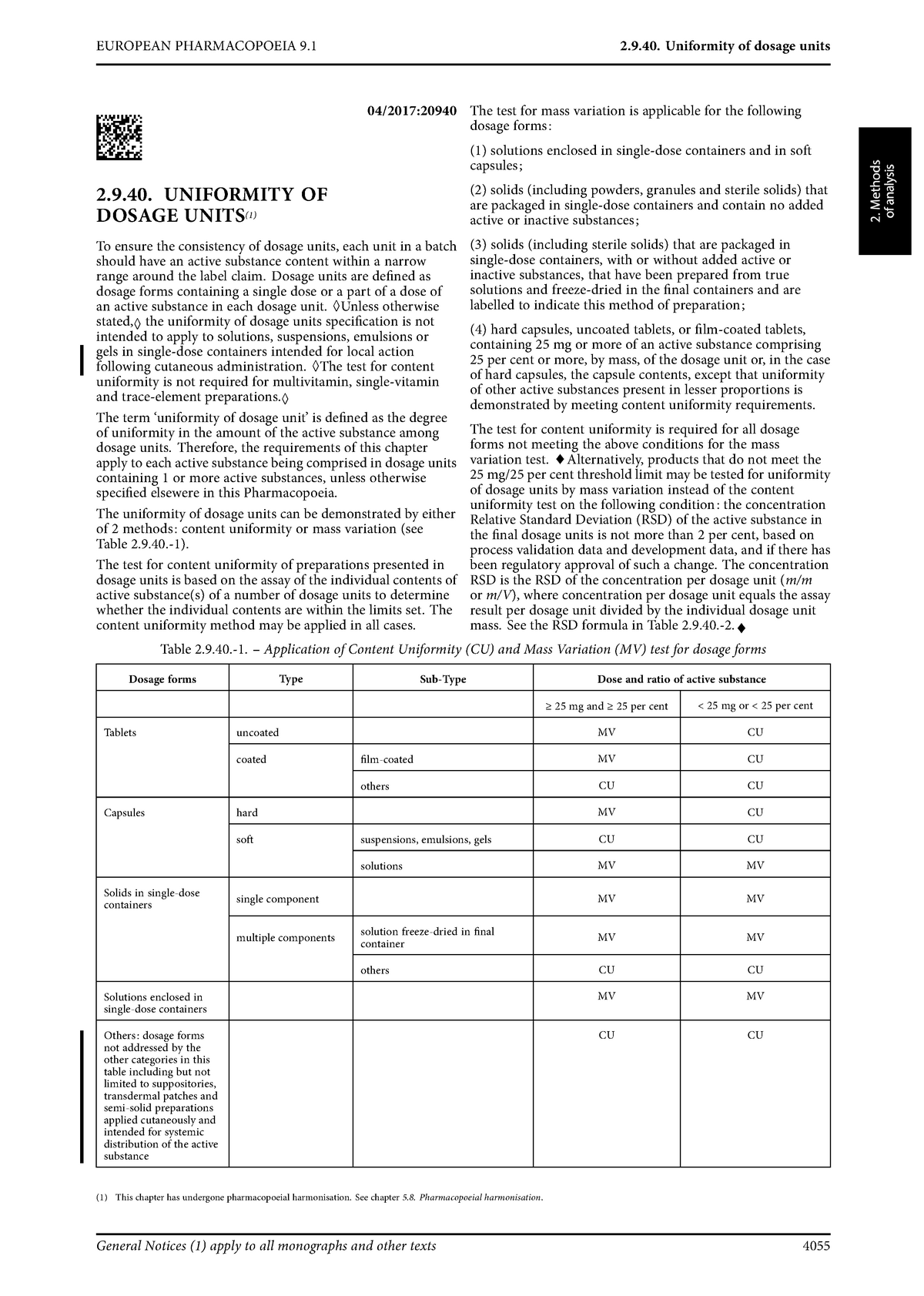
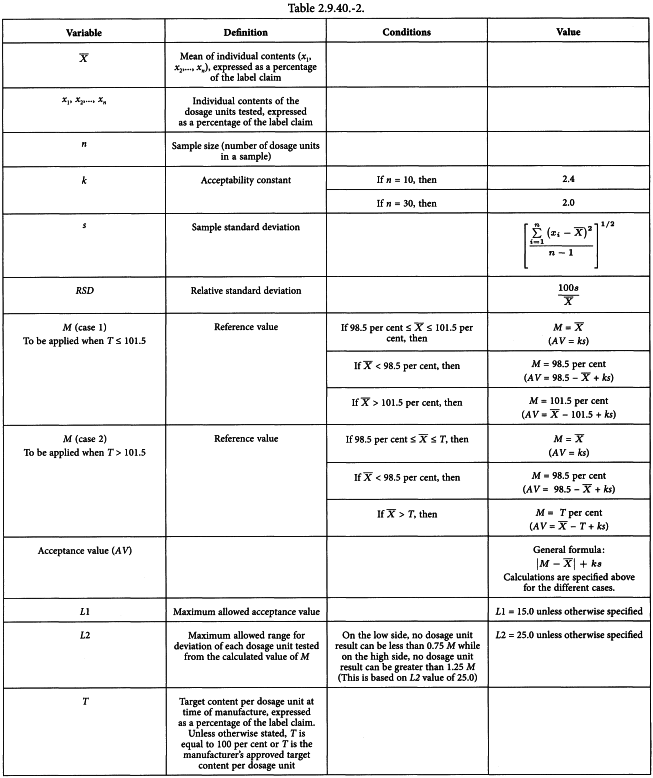
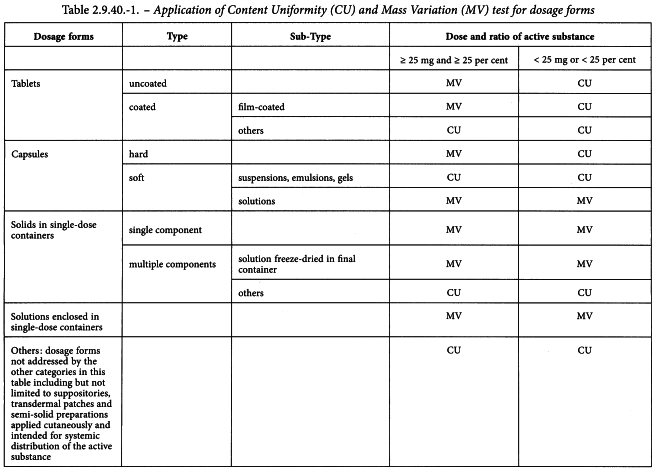
Uniformity of dosage units - EUROPEAN PHARMACOPOEIA 9 2.9. Uniformity of dosage units 04/2017: 2.9. - Studocu
![PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/555daba493a5301bf1879751f6ccef14b9a8c162/5-Figure3-1.png)
PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar

ASTM E2810-11 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units

ASTM E2810-11e2 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
![PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/555daba493a5301bf1879751f6ccef14b9a8c162/6-Figure4-1.png)
PDF] An Integrated Solution for Calculating Results from Tests for Content Uniformity of Dosage Units According to the United States and European Pharmacopeia | Semantic Scholar
ICH guideline Q4B annex 6 to note for evaluation and recommendation of pharmacopoeial texts for use in the ICH regions on unifor